The Pancreatic Cancer Action Network Urges All Pancreatic Cancer Patients to Consider Clinical Trial Enrollment
Pancreatic Cancer Patients Who Participate in Clinical Research Have Better Outcomes, yet Only 4.5 Percent of Patients Enroll
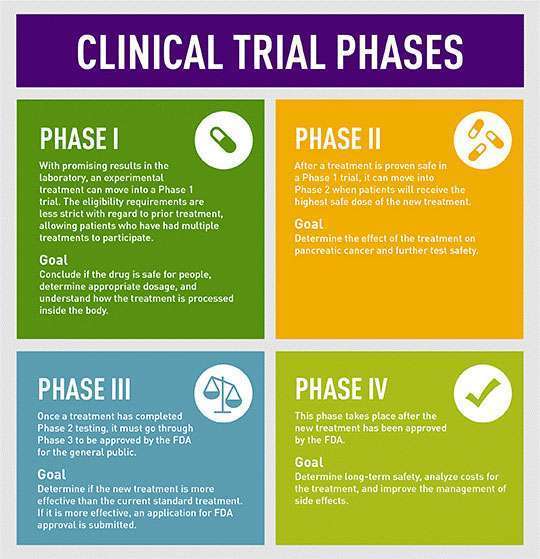
MANHATTAN BEACH, Calif. – (Jan. 10, 2018) Pancreatic cancer patients who participate in clinical research have better outcomes. So this January, National Pancreatic Cancer Clinical Trials Awareness Month, and throughout the year, the Pancreatic Cancer Action Network (PanCAN) urges all pancreatic cancer patients to consider clinical trial enrollment at diagnosis and during every treatment decision.
Without an effective early detection method and with few treatment options, an estimated 71 percent of pancreatic cancer patients will die in the first year of diagnosis. Only 9 percent of those diagnosed will survive five years. This survival rate remained unchanged over last year, according to the American Cancer Society’s Facts and Figures report, released Jan. 4. This underscores the importance of patients enrolling in clinical trials, which can lead to new treatments and improve outcomes.
Richard Blish, an eight-year pancreatic cancer survivor, is a perfect example of the importance of enrolling in a clinical trial. He credits his decision for his ability to fight the world’s toughest cancer.
“My diagnosis was at the cusp of stage I and stage II, giving me about a 30 percent chance of survival for five years,” said Blish. “However, my clinical trial (chemotherapy before Whipple surgery) which I participated in shrank my tumor by about 50 percent, allowing me to undergo a successful Whipple procedure, which improved my odds for survival several-fold.”
In the fight against pancreatic cancer, clinical trials provide the best treatment options, giving patients early access to cutting-edge treatments that can advance research, improve treatment options and increase survival rates.
“Patients who enroll in clinical trials at any stage in their diagnosis are not only helping themselves, but furthering advances in treatments needed to help countless others,” said Julie Fleshman, JD, MBA, president and CEO of PanCAN. “We maintain the most comprehensive and up-to-date database of pancreatic cancer clinical trials available in the U.S., which we use to help patients and healthcare professionals get the best information to inform treatment decisions.”
PanCAN Patient Services Case Managers can also provide, personalized clinical trials searches and answer questions about clinical trials. Case Managers can also provide comprehensive disease information, a list of specialists who treat a high volume of pancreatic cancer patients, access to precision medicine services, support resources and more.
Learn more about clinical trials by visiting pancan.org/clinicaltrials or by speaking with a PanCAN Patient Services Case Manager at 877-2-PANCAN. Follow the Pancreatic Cancer Action Network on Twitter, Instagram and Facebook.
















